|
8/31/2023 0 Comments Defects in crystal lattice 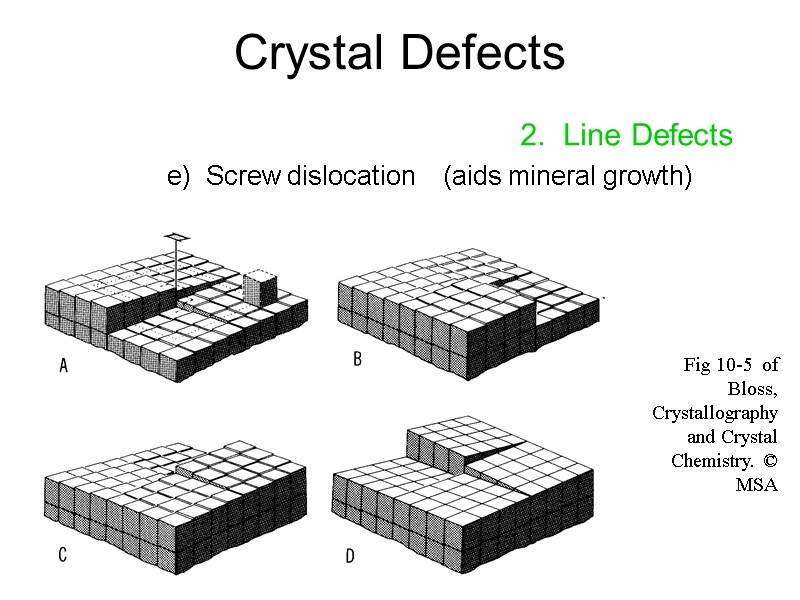
There could be di-vacancies (an association of two vacancies) or even tri-vacancies. 
The atoms surrounding a vacancy experience a slight displacement into the empty lattice site, and thus, a vacancy is a centre of approximately spherical distortion in the lattice, Fig. When an atom is missing from its lattice site in a crystal structure of a metal, it is called a vacancy (or vacant lattice site) as illustrated in Fig. A point defect extends its influence only a few atomic diameters beyond its lattice position. The size of the defect could be one atom, or two atomic diameters, which is just like a point. It is a defect of dimensions just like a point (zero dimensions). Impurity Atoms.Ī point defect is a very localised disruption in the regularity of a lattice. ISBN: 9780750646819.Various types of point defects are: 1. Chapter 5, Sample Problem 2, Practice Problem 2 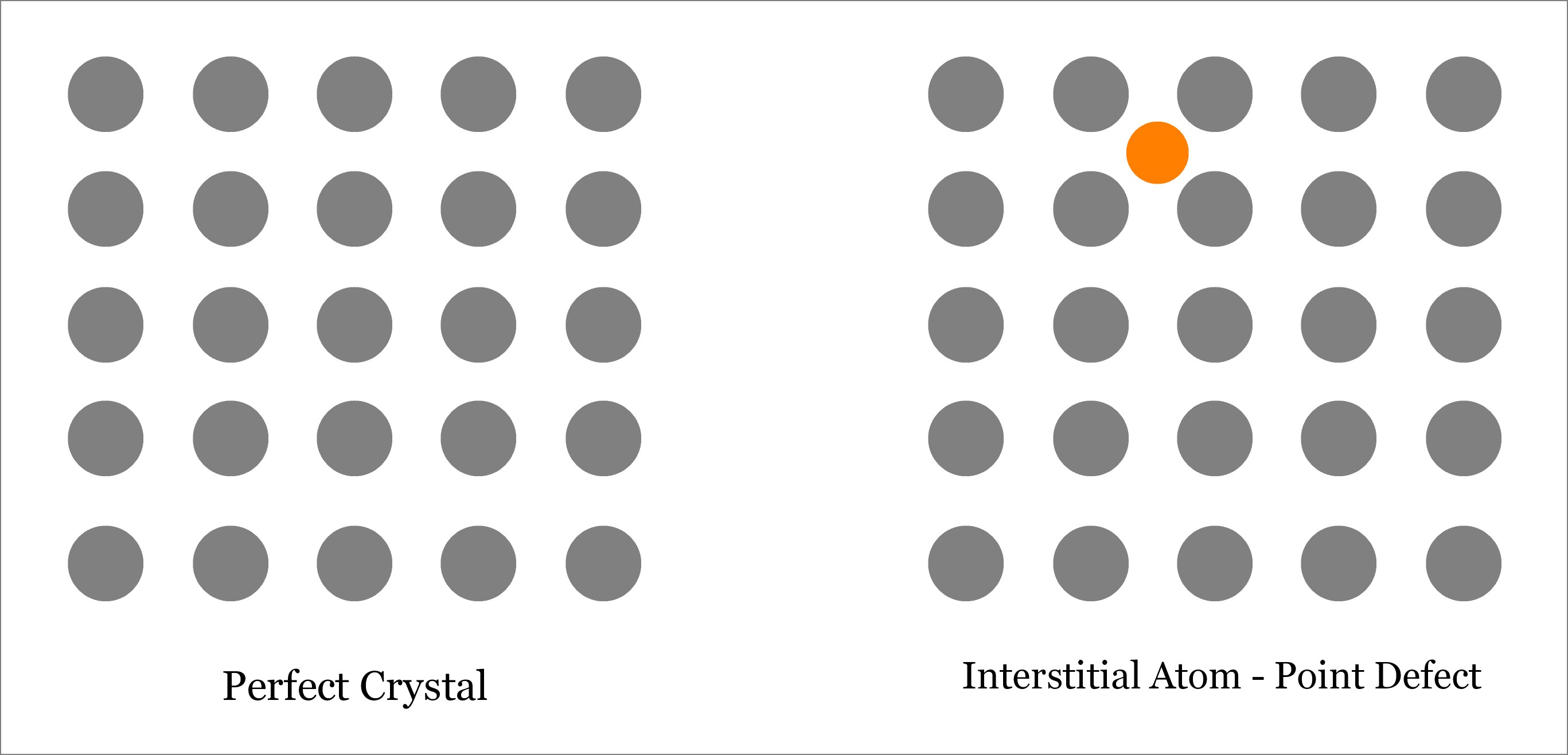
Line defects occur when a lattice mismatch runs through the crystal. In ionic crystals, overall charge neutrality must be preserved, so a whole stoichiometric unit may be removed to create two or more vacancies ( Schottky) one ion may move to an interstitial site ( Frenkel) or one or more electrons may fill an anionic vacancy ( F-center). P in Si, B in C), while interstitial sites can host atoms of A (self interstitial) or B (interstitial impurity, e.g. Vacancies in a regular lattice of A atoms may be filled by an atom of B ( substitutional, e.g. Creating an empty crystal lattice site ( vacancy) requires overcoming bonds with nearest-neighbor atoms, typically with thermal energy. Li + in saline solution (NaCl (aq)), others can create enhanced properties (e.g. While unwanted defects can weaken or contaminate materials (e.g. 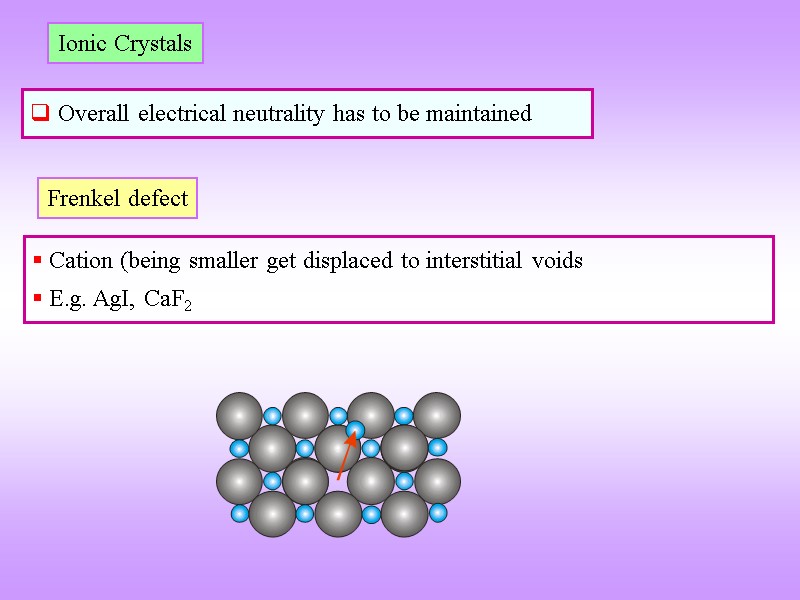
In the real world, materials rarely consist of single, perfect crystals defects in crystals occur naturally, or are introduced during processing. Michael Demkowicz ( homepage) lectures today, introducing the next topic: imperfections in crystal lattices. Lecture Slides (PDF - 3.0MB) Lecture Summary interstitials, Arrhenius plot, thermal expansion 5.2, “Thermal Production of Point Defects.”Īctivation energy of vacancies vs. 5.1, “Thermally-Activated Processes.”Īrrhenius equation, activation energy, Maxwell-Boltzmann distribution, process mechanisms and rate-limiting steps 4.3, “Linear Defects, or Dislocations – One-Dimensional Imperfections.”īurgers vector edge, screw, mixed, and partial dislocations Vacancies and interstitial defects, Schottky and Frenkel defects 4.2, “Point Defects – Zero-Dimensional Imperfections.” Random and ordered solid solutions, Hume-Rothery rules, interstitial and substitutional solutes, charge neutrality 4.1, “The Solid Solution – Chemical Imperfection.” Give 3 examples of additives that have a detrimental effect on the surrounding crystal, and 3 examples that improve the material properties.Īrchived Lecture Notes #6 (PDF), Sections 1-2ĭefects in metals, memory metal, defects in ionic and molecular crystals, nonstoichiometric compounds.Explain why a given crystal has either substitutional or interstitial impurity atoms.Write expressions for defects in a given ionic crystal, and explain why they have higher energies of formation than similar defects in metallic crystals.Calculate the vacancy concentration in a crystal at a given temperature.Sketch a crystal containing any of the following defects: substitutional impurity, interstitial impurity, vacancy, Schottky, Frenkel.Learning ObjectivesĪfter completing this session, you should be able to: Distribution of energies as described by Maxwell-Boltzmann statistics ( Session 14)Ĭompare the expression for the energy required to produce a vacancy, derived in this lecture, with the expression for the rate of a chemical reaction (the Arrhenius equation), presented in Session 22: Introduction to Kinetics.Point defect, line defect, substitutional impurity, interstitial impurity, vacancy, self interstitial, ionic defect, Hope Diamond, Schottky defect, Frenkel defect, F-center, charge neutrality, edge dislocation, screw dislocation, dislocation motion, bubble raft model, chemical imperfection, structural imperfection, formation energy, entropy factor, stoichiometric unit, effective charge, Kröger-Vink notationĪluminum (Al), steel, diamond, doped silicon, LaNi 5, copper (Cu), rock salt (NaCl), zirconia (ZrO 2)Īluminum alloys for soda cans, n-and p-type semiconductors, steel, hydrogen embrittlement of steel, Hope diamond, colored gold, hydrogen storageīefore starting this session, you should be familiar with: Point & Line Defectsĭefects in crystals: point defects, line defects 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
